Relentlessly Anti-Cancer
Primevax is leveraging powerful immuno-stimulatory properties of the dengue virus to prime the human body to fight late-stage cancers
Primevax is a clinical stage immuno-oncology company developing novel therapies for cancer. We are pioneering the world’s first use of the Dengue Virus as an oncolytic virotherapy to address late stage failed treatment markets.
We believe that the dengue virus presents a new method to invoke a strong anti-cancer immune response while demonstrating a high safety profile.
Primevax has patented the world’s only cancer treatment using dengue virus. Our lead asset PV001-DV, is a live, attenuated strain of Dengue Virus. This strain was originally developed by the U.S. Army as a vaccine, with extensive study in humans for over 20 years. The clinical studies show PV001-DV possesses an exceptional balance of safety and immunogenicity.
In recognizing this attractive anti-cancer profile, Primevax signed an exclusive license with the U.S. Army to develop DENV-1 45AZ5 for all cancers worldwide.
Our agreement also equips us to fully scale cGMP drug product for clinical studies, with a master file already filed with the FDA.
Dengue Virus (PV001-DV)
Dengue virus presents an opportunity to develop into a new first-in-class virotherapy.
The industry has witnessed multiple virotherapies develop, but few have ultimately translated into clinical benefit to cancer patients.
Unlike other oncolytic viruses, our dengue virus is not intended to be a genetically modified virus targeting a specific mutation or to serve as a backbone to deliver a particular antigen target.
Our naturally attenuated strain of dengue virus safely induces a native anti-dengue immune response in humans that is systemic in nature. And in the laboratory, we observe that this immune response results a combination effect of both direct oncolytic tumor killing as well as broad immunostimulatory effects which led to immune-mediated tumor killing. In total, we have observed at least 3 differentiated mechanisms of tumor killing.
PV001-DV’s data summary:
21 healthy volunteers treated: validated safety and powerful immune stimulation
Strong anti-tumor mechanisms of action repeatedly demonstrated in laboratory
Matching immune stimulation across healthy humans and cancer patient blood samples, further suggesting translatability of science to cancer patients
The above have been repeatedly demonstrated across multiple experiments, conditions, or tumor types.
We believe that to adequately address treatment-failed late stage cancer, the industry needs to overcome immune evasion and induce both local and systemic effects. In the totality of our publications, we observe that PV001-DV checks the right boxes to produce such powerful effects along multiple mechanisms both local and systemic.
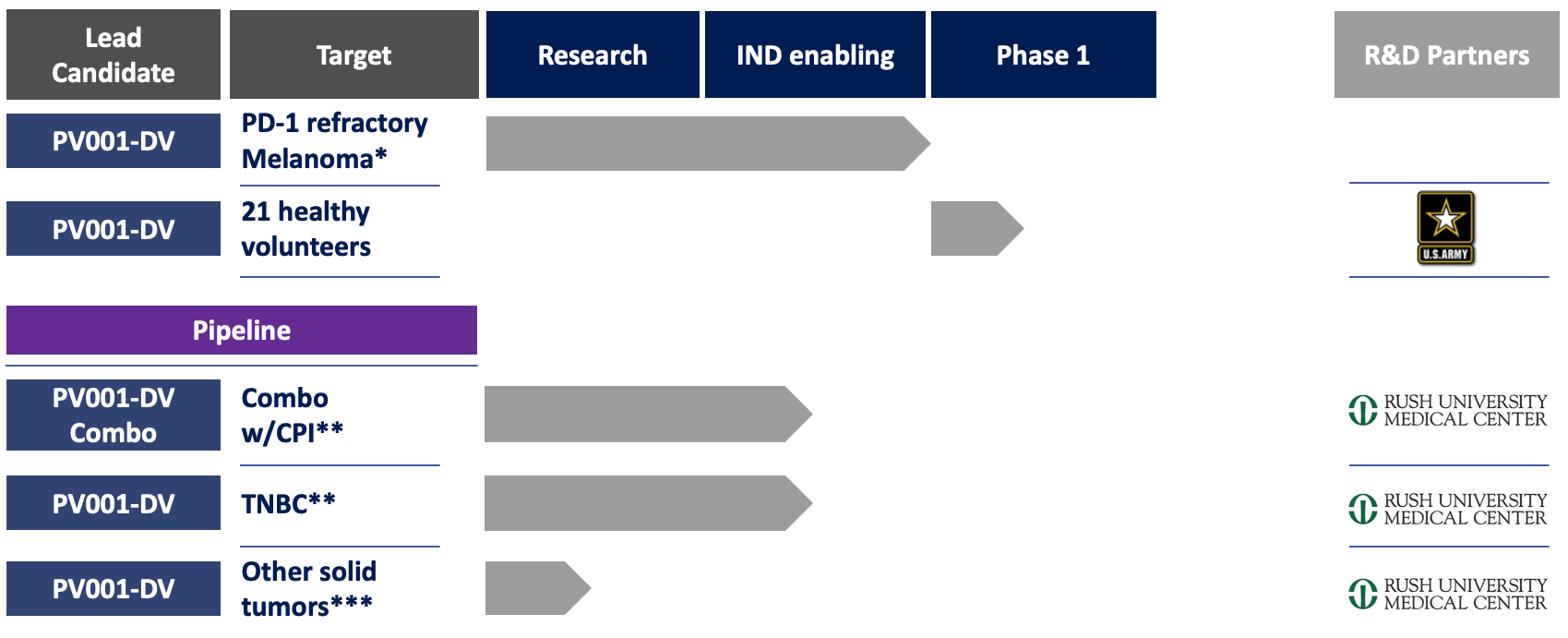
Pipeline
Our initial target is Melanoma with an IND open to treat PD-1 refractory patients for Phase 1b and Phase 2. Through our collaboration with the U.S. Army, we have Phase 1a safety studies in over 20 healthy volunteers. The clinical studies demonstrate PV001-DV has an outstanding safety profile and is well-tolerated. Our next stage is to enter the clinic for Phase 1b dose finding studies.
Our R&D pipeline includes preclinical studies in other solid tumors as well as combination with checkpoint immunotherapies.
* PD-1-Failed Melanoma: Patients with advanced melanoma who fail to respond to current best-in-class immunotherapies (PD-1 ± CTLA-4) ± iBraf
** Requires IND amendment
*** Solid Tumors: Patients with tumors that form as lumps in an organ or tissue of the body
Publications
Lead Papers
The below are primary papers supporting the safety and potential efficacy of using dengue virus to treat cancer. These papers summarize the tumor killing power of dengue, the clinical experience of the virus in humans, and immune activation biomarkers generated during human infections.
Other Dengue Resources
The below are additional secondary papers that support the immune potency or safety of the dengue virus from other researchers over the last 20 years. The dengue virus is well characterized in the literature, and it is shown to have powerful immune activation effects while consistently demonstrating safety.
Taiwan epidemic study: dengue and immunosuppressed cancer patients
CD8+ T cell expansion and activation in dengue
CTL home to the skin during dengue infection
Bystander T-cell activation in Dengue